Cephalopod Biochemistry
Our group studies the chemical and physical origins of biological color in dynamic systems like cephalopods, including squid, octopus, and cuttlefish. Our primary focus is on the role of dermal chromatophore organs as active components of color change and their function in facilitating camouflage, where we study the pigmented nanostructures localized within chromatophores and more recently the musculoskeletal system surrounding these nanostructures that supports motility and signaling.
Cephalopods behave as living adaptive sensors and displays evolved to facilitate a signaling feedback loop (reading, writing, and scrambling optical signature) that leads to distributed body patterning during camouflage. Our group is working to answer (i) what are the structural components of the chromatophore? and (ii) how do they contribute to signal propagation? Our goal is to use our learnings to create a biomolecular blueprint of the cephalopod chromatophore organs to guide the design of future color changing systems.
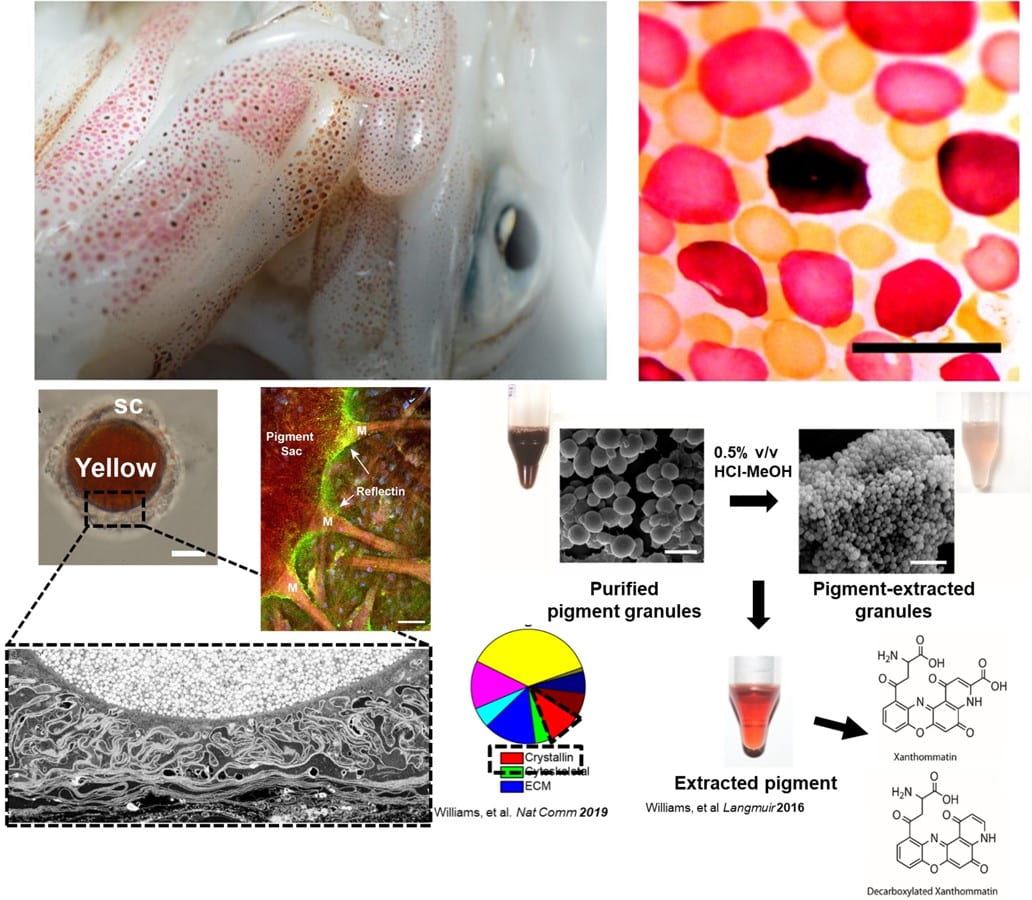
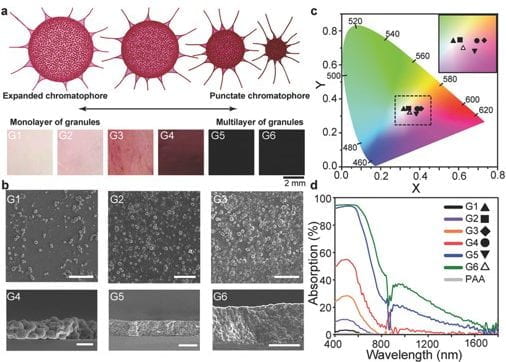
Beginning with the first mass spectrometric analysis of squid chromatophore tissue, we identified the abundance of phenoxazone-based chromophores (xanthommatin, Xa, and its decarboxylated form, DC-Xa) that combine to contribute to a broad-range of color spanning the UV-visible spectra. Our lab worked with a team of researchers across 4 institutions to compile the first annotated chromatophore proteome, which was used to describe the biochemical origins of both pigmentary and newly discovered structural color emanating in precise register in squid chromatophores. This exciting finding highlighted an extremely rare case of how pigments and protein-based nanostructures are tightly co-localized within a single, organ to give rise to rapid (~100 msec) changes in color. We expanded this finding with computational analyses supported by mass spectrometry to show that the Xa pigments are confined to nanostructured granules composed of lens crystallin proteins in the chromatophore that we describe as having dual roles: stabilizing the charge environment of the absorptive pigment molecules and simultaneously enabling optical scattering that could aid in color amplification during actuation. These findings suggest a functional convergence between the eyes and skin of the animals that is the subject of ongoing studies.
We have also extrapolated the complex refractive index (RI) from experimentally determined real and approximated imaginary portions of the cephalopod pigments to better understand their optical function in the dynamic chromatophores. Based on our experimental results, we determined the complex RI of the pigments with a real portion (n) of 1.66 (±0. 05) – 1.92 (±0. 11) and an imaginary portion (k) of 0.13 (±0.08)i. These values are high for biological molecules; where, the closest comparators are experimentally derived and approximated natural and synthetic melanin with an n range of 1.7-2.0 and naturally occurring guanine crystals in copepods, which have an RI of 1.83.
.